 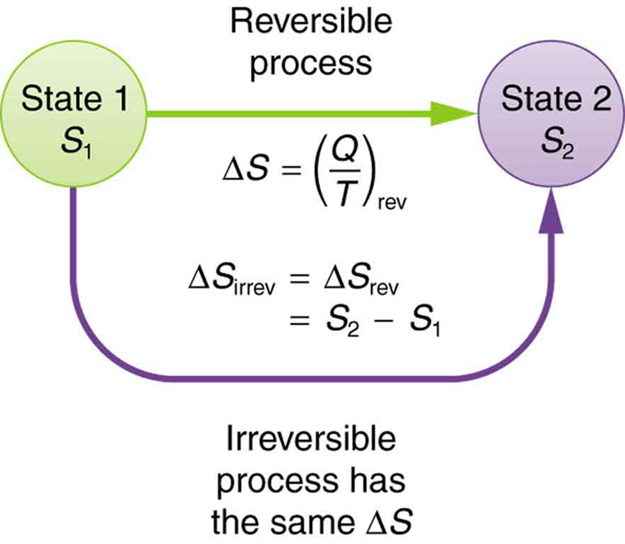
The fact that the absolute value of specific entropy is unknown is not a problem because it is the change in specific entropy (Δs) and not the absolute value that is important in practical problems. Not because the equation itself is that confusing(it isnt. For example, the specific entropy of water or steam is given using the reference that the specific entropy of water is zero at 32☏. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s. Boltzmanns entropy formula is possibly the one of the most difficult equations in Physics. Also, like enthalpy, the entropy of a substance is given with respect to some reference value. Like enthalpy, entropy cannot be measured directly. In one statistical interpretation of entropy, it is found that for a very large system in thermodynamic equilibrium, entropy S is proportional to the natural logarithm of a quantity representing the maximum number of microscopic ways in which the macroscopic state corresponding to S can be realized that is, S k ln. For example, the entropy of a solid, where the particles are not. Entropy is represented by the letter S and can be defined as ΔS in the following relationships. Entropy is a thermodynamic function used to measure the randomness or disorder of a system. 
Entropy is sometimes referred to as a measure of the inability to do work for a given heat transferred. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s = S/m) as part of the information tabulated. Entropy quantifies the energy of a substance that is no longer available to perform useful work. Because entropy is a property, changes in it can be determined by knowing the initial and final conditions of a substance. H -92.6kJ/mol Solution We can use the formula S 2 (NH3) (N2) + 3 (H2) S (2) (192.5 JK-1mol-1) 191.6JK-1mol-1 + (3) (130.6 JK-1mol-1) S -198. > Thermodynamics Directory | Heat Transfer DirectoryĮntropy Definition - Thermodynamic PropertiesĮntropy (S) is a property of a substance, as are pressure, temperature, volume, and enthalpy. Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic quantities. The fact that a perfect crystal of a substance at 0 K has zero entropy is sometimes called the Third Law of Thermodynamics.Entropy Definition and Equation Thermodynamics Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system. This is because we know that the substance has zero entropy as a perfect crystal at 0 K there is no comparable zero for enthalpy. The entropy of a system is related to its energy and temperature, as stated by the equation S Q/T, where S is the change in entropy, Q is the heat added. The reason is that the entropies listed are absolute, rather than relative to some arbitrary standard like enthalpy. Note that there are values listed for elements, unlike DH fº values for elements. The Thermodynamics Table lists the entropies of some substances at 25 ✬. Continue this process until you reach the temperature for which you want to know the entropy of a substance (25 ✬ is a common temperature for reporting the entropy of a substance). Then you can use equation (1) to calculate the entropy changes. Even though equation (1) only works when the temperature is constant, it is approximately correct when the temperature change is small. Now start introducing small amounts of heat and measuring the temperature change. Since there is no disorder in this state, the entropy can be defined as zero. Imagine cooling the substance to absolute zero and forming a perfect crystal (no holes, all the atoms in their exact place in the crystal lattice). We can then determine the value of the specific heat. The absolute entropy of any substance can be calculated using equation (1) in the following way. If we have a constant volume process, the second term in the equation is equal to zero, since v2/v1 1. At absolute 0 (0 K), all atomic motion ceases and the disorder in a substance is zero. On this scale, zero is the theoretically lowest possible temperature that any substance can reach. The temperature in this equation must be measured on the absolute, or Kelvin temperature scale. Using this equation it is possible to measure entropy changes using a calorimeter. Where S represents entropy, DS represents the change in entropy, q represents heat transfer, and T is the temperature. 
(Here monatomic means that we approximate the atoms by point particles, and ideal means that those particles do not interact with each other. One useful way of measuring entropy is by the following equation: We do so by using our statistical definition of entropy to calculate the entropy of a monatomic ideal gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
